TECHNOLOGICAL UPDATES IN OPHTHALMOLOGY
Use of vital dyes in vitreo-retinal surgery
MONTERO MORENO JA1, RUIZ MORENO JM2, FERNÁNDEZ MUÑOZ M3
1
Ph.D. in Medicine and Surgery. Retina-Vitreous Unit, Alicante Ophthalmology
Institute. VISSUM. Ophthalmology Service. Pío del Río Hortega Hospital.
Valladolid. Spain.
2
Ph.D. in Medicine and Surgery. Ophthalmolgy Professor. Castilla La Mancha
University. Albacete. Spain. Retina Vitreous Unit. Alicante Ophthalmology
Institute. VISSUM.
3
Ph.D. in Medicine and Surgery. Ophthalmolgy Service. Río Carrión Hospital.
Palencia. Spain.
ABSTRACT
In recent years, surgical techniques assisted by the use of vital dyes have become popular in endo-ocular surgery as they facilitate procedures in adverse conditions such as the case of capsulorrhexis in mature cataracts or allow the execution of new techniques such as internal limiting membrane peeling. This article reviews the indications, applications and characteristics of vital dyes in vitreo-retinal surgery.
Key words: Vitrectomy, epi-retinal membrane, internal limiting membrane, indocyanine green, infracyanine green, brilliant blue, triphan blue, triamcionolone acetonide.
The term «chromo-vitrectomy» has been coined to define the use of vital dyes (i.e., staining able to modify the color of live tissue causing minimum damage to the cells) in vitreo-retinal surgery. The usefulness of vital dyes (VD) lies in their specific ability to stain different structures such as cells or collagen fibers. In addition to its use in vitreo-retinal surgery, VD has been utilized in other ophthalmological fields such as surgery of the cornea or conjunctiva, glaucoma, cataracts, strabismus, orbit and lachrymal pathways.
The main indication for chromovitrectomy is for staining the transparent structures of the vitreo-retinal interphase. This procedure was first mentioned in the late seventies when Abrams mentioned the application of sodium fluorescein for identifying the vitreous. This type of procedure was not brought up again until the early days of this century, when it was considered that a better definition of tissue could reduce the associated tissue damage (particularly on the retina nervous fiber layer) after the first results published by Eckardt in the internal limiting membrane (ILM) peeling for macular hole treatment. Since then, the success rate in the closure of macular holes reached up to 96% against the previous 58-95% described by some authors. To achieve this, ophthalmological intravenous dyes were utilized (fluorescein and indocyanine green, ICG), utilizing the properties of triamcinolone for staining the vitreous. Said properties had already been observed after injections by Peyman and subsequently applied by Burk for staining the anterior chamber-prolapsed vitreous.
The surgical peeling of the ILM could damage the nervous fiber layer (with the ensuing visual field defect described in up to 50% of cases) and damage the retina pigmentary epithelium (RPE). Post-surgery campimetric defects have been related to the surgical trauma suffered by the papilla, to the liquid-gas exchange (due to tissue dehydration), with raised post-op intraocular pressure and direct trauma of the retina.
Both the surgical trauma and phototoxicity can cause RPE damage which expresses in the form of hyper pigmentation or hypofluorescence in AFG.
TECHNIQUE FOR APPLYING THE DYE DURING VITRECTOMY
Various techniques for staining the vitreous cavity with vital dyes have been described. One is the so-called «dry technique» which is carried out with the eye full of air after removing the liquid from the vitreous cavity followed by a liquid-gas exchange before injecting the dye. The advantage of this procedure is a higher concentration of dye over the retina and avoiding contact with the lens, against the disadvantage of inducing possible retinal toxicity due to its high concentration.
The second procedure is the so-called «wet method» carried out with the eye full of liquid (generally a balanced saline) while the dye is injected over the retinal surface. In these cases, the dye concentration is lower because it is diluted in the vitreous cavity fluid, the drawback being that the dye could disperse and stain other retinal areas or the posterior lens. Czajka et al compared both procedures in a pig eye model and found that the first procedure was associated to a greater prevalence of RPE atrophy and external retina degeneration.
Harbin and Chu developed a dye application system called VINCE (Vitreoretinal Internal limiting membrane Color Enhancer; Dutch Ophthalmic, Zuidland, The Netherlands), that consisted in a brush comprising a 20 G modified reflux cannule containing a silicone tube surrounded by a metal cannule. This brush allows to paint the dry directly over the tissue for enhanced visualization while avoiding uncontrolled staining of the macular hole RPE and the peripheral retina.
In order to avoid contact between the dye and the RPE after passing through the macular hole it was proposed to make slow injections utilizing devices such as VINCE, or placing a bubble of liquid over the hole (liquid perfluorocarbon, autologous whole blood and sodium hyaluronate). Liquid perfluorocarbon (LPFC) as protective agent for the RPE in the macular hole when staining with ICG as both do not mix. Scupola et al communicated the results after 1 year of protecting the RPE with LPFC, without observing defects at the RPE level and with a mean final visual acuity of 20/50. However, the results included an increase in the cost of surgery and surgery times as well as the necessity of withdrawing the LPFC completely to avoid retinal toxicity. Lai et at, after the use of autologous blood for protecting the RPE from ICG, observed that the cellular damage was reduced and no differences could be found between the eyes treated with whole blood, plasma or hematite concentrates, without evidencing tissue damages or remains of ICG after 1 month of follow-up.
VITAL DYES IN CHROMOVITRECTOMY
Even though all authors agree that the use of VD improves the visualization of pre-retinal structures, several factors make it advisable to be prudent in their use. These are:
1. Mismatch between the functional and anatomic results of macular hole surgery. There is no agreement about the possible toxicity of the ICG, possibly due to the greater care taken in the surgical use of ICG.
2. The bonding mechanism between ICG and triphan blue (TB) on tissue is not clear. It has been proposed that ICG could exhibit high affinity with extra-cellular structures of ILM such as type IV collagen, laminin and fibronectin, while TB would stain the epiretinal membranes (ERM) more than the ILM.
3. New instruments such as VINCE should allow limited tissue staining.
4. The toxicity mechanism of ICG on the retina is not clear: it could be due to ICG or its metabolites, or to the alteration of the vitreo-retinal interphase osmolarity. Another possible toxicity mechanism could be the iatrogenous trauma of the peeling itself, as suggested by the associated campimetric defects encountered in the post-op. ICG has a photosensitizing effect, which could include tissue damage by raising the local temperature (type I photo oxidation) or by a photodynamic mechanism (type 2 photo oxidation).
Indocyanine green (fig. 1)
Cyanines include a -CH= group joined to two heterocyclic rings with nitrogen belonging to a larger group, that of polymethylic dyes. Cyanines are highly colored organic compounds which have been utilized mainly in photography and the textile industry.

Fig. 1: Commercial presentation of liophylized indocyanine
green (ICG).
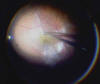
ICG is an anionic vital dye having the theoretical formula C43H47N2NaO6S2 (although it includes about 5% of iodine derived from the synthesis process), and a molecular weight of 775 daltons. It bonds both to cell elements (ERM, fig. 2) as non-cell elements (ILM). It is usually in lyophilized sterile form ready for dissolving in distilled water. In 1959 it was approved by the FDA as an indicator of bodily dissolutions and in liver function tests. ICG is utilized in angiography because it allows the study of choroidal circulation, with the absorption and fluorescence peaks being found in the near infrared spectrum.

Fig. 2: Staining and peeling of the epi-retinal membrane (ERM)
after ICG injection.
In the field of ophthalmological surgery and despite its popularity it is used without indication. It can be found under different brands such as ICG-Pulsion (Pulsion Medical Systems; 25mg and 50 mg vials); ICV Indocianina Verde (Ophthalmos; 5, 25, and 50 mg vials); Diagnogreen (Daiichi Pharmaceutical; 25 mg vial); and IC-Green (Akorn Inc.; 25 mg vial).
The end purpose of the dilution is to obtain a concentration between 0.05 and 0.5%, initially with distilled water, to be continued with saline due to the greater risk of precipitation of the latter.
Tests in pig models determined that, after exposure to light, ICG increases the biochemical rigidity of ILM, thus facilitating its peeling. A vast amount of papers have been published describing ILM peeling as less traumatic than peeling without staining, and with good clinical results, achieving between 74% and 100% of macular hole closures after peeling with ICG (figs. 3-4). In diabetic macular edema cases, the utilization of ICG in ILM peeling has been followed by visual improvements. However, in persistent macular edema after occlusion of the central retinal vein and Irvine-Gass syndrome, no significant clinical improvement has been observed.

Fig. 3: Internal Limiting membrane (ILM) staining and peeling
in a macular hole case after ICG injection.

Fig. 4: ILM staining and peeling Tinción in a case of macular
hole after ICG injection.
The drawback of ICG is its possible toxicity; it can remain in the tissues up to 36 months after the intervention and migrate to the sub-retinal space through the macular hole. The literature describes alterations in the RPE and campimetric and papillary defects.
Recent studies on the visual results of ILM peeling with ICG staining could be more favorable than previous studies due to incubation time changes (from 0 to 5 minutes; RPE defects appear after 30 seconds) and in the concentration (toxicity for RPE is greater when the osmolarity of ICG is under 270 mOsm and its concentration is over 0.5%). After observing defects in the RPE and in the visual field in the first cases of use of ICG at a concentration of 0.5%, it was proposed to reduce its concentration to 0.125%. However, at this concentration published results vary widely: some authors continue to find over 50% of cases with toxicity for RPE, attributed to osmolarity differences, dye application techniques and exposure to light. Recently ICG has been utilized at a concentration of 0.05% with osmolarity close to 290 mOsm without hardly any effects on the RPE and with results similar to those of peeling without staining.
Histological studies made on the material removed after ILM peeling with ICG staining suggest that the cleavage plane is actually deeper than it was thought, as Müller cells have been found adhered to the ILM. It has been postulated that the damage described at the level of the RPE and of other retina cells could be due to contact with ICG followed by exposure to wavelengths above 620 nm, and that this effect is dosage-dependent.
It has been proposed to use ICG staining in ERM surgery for diabetic proliferative vitreoretinopathy, in idiopathic ERM and in proliferative vitreoretinopathy (PVR). The staining capacity of ICG if much higher on the acellular ILM than on the ERM, which on some occasions could be beneficial due to this negative staining.
Indocyanine green
Indocyanine green is a green dye having the same formula and characteristics as ICG. Infracyanine green (IfCG) is marketed as Infracyanine (Laboratoires SERB, 25 mg vials). Its difference with ICG lies in that it does not contain sodium iodide, which allows it to be dissolved in 5% glucose serum. It is an advantageous system for staining pre-retinal tissues and membranes as it avoids the exposure of the retina to iodine. As with ICG, IfGG is able to bond with non-cell ILM, but does not bond easily to epiretinal membranes. Its toxicity has not been demonstrated.
IfGG must be dissolved in 5% glucose serum to produce an iso-osmotic solution of 294-314 mmol/kg.
Good anatomic and visual results have been obtained after IfGG-assisted ILM peeling in macular holes and diabetic macular edema cases. The immunohistochemical study of the extracted material exhibits a positive stain for the glyofibrilar acid protein (GFAP) and S-100, which indicates the presence of cellular remains of Müller and ganglionary cells respectively, and could explain why in cadaver eyes IfGG and ICG staining followed by illumination is able to alter the cleavage plane of internal retina surgery. At the clinical level this fact can be associated to alterations in the RPE and post-op visual fields. Even so, IfGG seems to offer a safer profile for chromovitrectomy than IGC. In addition, it allows the identification if ILM at a concentration of 0.5 mg/ml with less toxic effects.
Sodium Fluorescein (fig. 5)
Sodium fluorescein belongs to the xanthene group, made up of yellow heterocyclic organic compounds having the formula C13H10O such as eosin and rhodamin. This group of dyes has a fluorescence from yellow to pink or from bluish to red. The fluorescein formula is C20H12O5 and its molecular weight of 332 daltons. It exhibits marked fluorescence with an absorption peak of 494 nm and maximum emission of 521 nm. Fluorescein can be admixed with different salts such as sodium fluorescein or diacetate. In both forms, it is able to efficiently stain the vitreous to facilitate its complete removal in posterior vitrectomy or in the prolapse thereof into the anterior chamber after cataract surgery complications.

Fig. 5: Commercial presentation of sodium fluorescein
dissolution.
Triphan Blue (figs. 6-8)
Azoic dyes constitute a range of synthetic dyes containing nitrogen in azoic form (-N5N-) which connects two aromatic rings. These dyes have brilliant, high intensity colors which can be easily modified and can be found at very affordable prices.

Fig. 6: Commercial presentation of Triphan Blue dissolution
(Membrane Blue) for ERM staining.
TB is a hydrophilic anionic azoic dye having the formula C34H24N6Na4O14S4 and a molecular weight of 960 daltons. It has been broadly used in vitrectomy and in cataract surgery. It can be obtained at a concentration of 0.15% for vitreous surgery under the Membrane Blue brand (DORC International, Zuidland, The Netherlands) and as Vision Blue at a concentration of 0.06% for cataract surgery (DORC).

Fig. 7: ERM staining with Triphan blue.
Both presentations include a formula comprising sodium salts, 8.2 mg of NaCl and water with an osmolarity between 257 and 314 mOsm/Kg, with a pH ranging between 7.7 and 7.6 TB exhibiting high affinity for ERM due to its dead glyal cell content and the fact that it does not stain the ILM as well as ICG. However, a comparative study on the ILM peeling ease in macular hole cases with ICG and TB, as well as the hole closure percentages, has demonstrated that the anatomic closure percentages were similar and that the visual result was significantly better in eyes stained with TB.
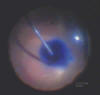
Fig. 8: ERM staining and peeling after Triphan blue injection.
In order to improve the penetration of dye into the surface it is possible to mix the solution with glucosylated serum at 5-10% (higher concentrations of glucose must be avoided due to the risk of producing severe hyperosmolar toxicity on the retina) in order to increase the dye density and allow its use in eyes with saline solution and avoid the liquid-air exchange.
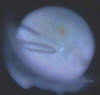
Fig. 9: Commercial presentation of brilliant blue dissolution
(Brilliant Peel) for staining the ILM.
Many researchers agree that this is an adequate dye for staining and eliminating ERM because it stains its entire surface in addition to reducing the retinal trauma during removal and facilitates the identification of its entire surface.
Most studies observed the absence of toxicity for the retina and the RPE, although one case was reported in which the dye migrated to the sub-retinal space, producing changes in the RPE without visual repercussions. The histopathology of extracted tissue did not show the presence of retinal cells or apoptotic tissue. Multifocal electroretinography did not reveal damages either, while electronic microscopy showed ILM remains adhered to the ERM, which also occurs when no dyes are utilized.
Brilliant Blue (figs. 9 and 10)
Brilliant Blue (BriB), also known as Acid Blue or Coomassie, is an anionic dye of the amino triarylmethane group having the empirical formula C47H48N3S2O7Na and a molecular weight of 854 dalton. It is utilized as a food dye and has been utilized without indication for staining the capsule and in vitrectomy. It was approved in the European Union in 2007 as Brilliant peel (Fluoron/Geuder, Heidelberg, Germany).

Fig. 10: ILM staining and peeling in a macular hole after
injection of brilliant blue in 5% dextrose.
In the human eye, an adequate staining is achieved in the ILM utilizing BriB as an iso-osmolar solution at a concentration of 0.25 mg/mL, without toxic effects on multifocal ERG being described (figs. 9 and 10).

Fig. 11: ERM peeling after intravitreal injection of
triamcinolone acetonide.
Patent Blue
Patent Blue (PB) is a hydrophilic dye of the anionic triaryl methane group, having an empirical formula of C27H31N2NaO6S2 and a molecular weight of 582 daltons. In it sodium or calcium salt form, it is utilized
As a fluorescent indicator to identify fungi in vitro. Since 2003 its use is authorized in the European Union for staining the capsule during cataracts surgery at a concentration of 0.24% under the trade name of Blueron (Geuder). In addition, it has been utilized without indication in vitrectomy.
Experimental studies and preliminary results of clinical research have demonstrated a moderate affinity of PB with ERM and the vitreous, and low affinity with ILM, although in the clinic it has been seen that this vital dye is as adequate as TB for staining the ERM. There is no agreement concerning its possible toxicity because, even though some studies describe moderate and reversible toxicity at the retinal level, in vitro studies demonstrate no toxicity for RPE even after sub-retinal injection. However, the PB intravitreous injection safe dose is not year clear.
Corticoids
Corticoids are a group of hormones which, in addition to being produced naturally in the supra-renal cortex, can also be synthesized. Corticoids are frequently used in clinics. Triamcinolone acetonide (TA)is a synthetic corticoid insoluble in water having the empirical formula C24H31FO6 and a molecular weight of 434 daltons. Fluometholone acetate (FMA) is a fluorinated glucocorticoid having the formula C24H31FO5 and a molecular weight of 418 daltons. Both corticoids can be utilized in surgery for their anti-inflammatory effect as well as for staining the vitreous by means of depositing crystals on it. TA is marketed under the trade name of Triesence (Alcon Labs, Forth Worth, TX, 40 mg/mL); Kenalog (Bristol-Myers-Squibb, Peapack, NJ, 40 mg/mL); Trivaris (Allergan, Irvine, CA, 80 mg/mL); and in Spain as Trigon Depot (Squibb, 40 mg/mL).
Kimura et al first utilized TA for peeling ILM as a result of the crystals sedimentation over the ILM which facilitated its identification and elimination with good clinical results and without adverse effects 3 months later (fig. 11). Comparing these results with those of ICG staining in macular hole cases, a similar rate of closures without adverse effects have been described.
Bromophenol Blue
The use of bromophenol blue (BroB) has been proposed as an alternative for chromovitrectomy. A comparative study of six vital dyes (BroB, Clear Green Yellow, E68, Chicago Blue, Rhodamine, Basic Rhodoline Blue and Basic Rhodoline Blue 3) found that the ILM and ERM staining were better with BroB and that no damages were caused to the RPE or proliferation thereof in vitro in concentrations between 0.2% and 0.02%. In addition, it was found that its utilization in vivo in rat and pig eyes at concentrations ranging between 0.5% and 0.02% included less retinal toxicity from the histological viewpoint and of the ganglionary cell count than the rest of mentioned dyes. Furthermore, at concentrations of 1%-2% it facilitates ILM staining and identification, although its utilization in the clinic is not yet established.
OTHER APPLICATIONS OF CHROMOVITRECTOMY
Sub-retinal injection for visualizing retinal tears
It is estimated that in 2.2-4% of retina detachments in phakic eyes and 7-16% of aphakic eyes, the tear location cannot be discovered. In 1939 the Kiton-Fast-V-Green IV injection was utilized for detecting tears because the retina stains green while the tear remains unstained. In the fifties IV fluorescein was utilized for the same purpose. In 1947, Black injected methylene blue through the sclera without success. More recently, Jackson applied 0.15% TB with a 41-G cannula during surgery to identify retinal tears in 4 out of 5 cases without detecting retinal toxicity.
Vital staining of the vitreous during vitrectomy
The full elimination of the vitreous improves surgical results in macular holes, macular edema and diabetic retinopathy.
The deposit of TA crystals over the acellular tissue enhances contrast between the areas with vitreous and those without it. The technique only consists in injecting TA in the vitreous cavity, which seems to contribute to reducing the frequency of fibrinoid and vitreo-retinal proliferation reactions, although it increases the risk of glaucoma.
Sodium fluorescein is very well absorbed by the vitreous and, when injected in the vitreous cavity at a concentration of 0.6%, it enhances the visibility of the vitreous without complications, even being inferior to TA. Fluometholone acetate can be a safe alternative for TA.
The intra-chamber or intra-vitreous use of TB can assist in detecting the vitreous prolapsed into the anterior chamber or the remains thereof in the vitreous cavity but with lower visibility than TA or fluorescein.
The future goals of chromovitrectomy include choroidal neovascular membranes, prematurity retinopathy, proliferative vitreoretinopathy and intra-ocular tumors.
REFERENCES
-
Rodrigues EB, Meyer CH, Kroll P. Chromovitrectomy: a new field in vitreoretinal surgery. Graefes Arch Clin Exp Ophthalmol. 2005 Apr; 243(4): 291-3.
-
Mennel S, Meyer CH, Tietjen A, Rodrigues EB, Schmidt JC. Patent blue: a novel vital dye in vitreoretinal surgery. Ophthalmologica. 2006; 220(3): 190-3.
-
Rodrigues EB, Maia M, Meyer CH, Penha FM, Dib E, Farah ME. Vital dyes for chromovitrectomy. Curr Opin Ophthalmol. 2007 May; 18(3): 179-87.
-
Rodrigues EB, Meyer CH, Mennel S, Farah ME. Mechanisms of intravitreal toxicity of indocyanine green dye: implications for chromovitrectomy. Retina. 2007 Sep; 27(7): 958-70.
-
Farah ME, Maia M, Furlani B, Bottos J, Meyer CH, Lima V, et al. Current concepts of trypan blue in chromovitrectomy. Dev Ophthalmol. 2008; 42: 91-100.
-
Haritoglou C, Schuttauf F, Gandorfer A, Thaler S. An experimental approach towards novel dyes for intraocular surgery. Dev Ophthalmol. 2008; 42: 141-52.
-
Rodrigues EB, Meyer CH. Meta-analysis of chromovitrectomy with indocyanine green in macular hole surgery. Ophthalmologica. 2008; 222(2): 123-9.
-
Rodrigues EB, Penha FM, Furlani B, Meyer CH, Maia M, Farah ME. Historical aspects and evolution of the application of vital dyes in vitreoretinal surgery and chromovitrectomy. Dev Ophthalmol. 2008; 42: 29-34.
-
Costa Ede P, Rodrigues EB, Farah ME, Dib E, Penha F, Magalhaes O, Jr., et al. Vital dyes and light sources for chromovitrectomy: comparative assessment of osmolarity, pH, and spectrophotometry. Invest Ophthalmol Vis Sci. 2009 Jan; 50(1): 385-91.
-
Farah ME, Maia M, Rodrigues EB. Dyes in ocular surgery: principles for use in chromovitrectomy. Am J Ophthalmol. 2009 Sep; 148(3): 332-40.
-
Henrich PB, Haritoglou C, Meyer P, Ferreira PR, Schotzau A, Katamay R, et al. Anatomical and functional outcome in brilliant blue G assisted chromovitrectomy. Acta Ophthalmol. 2009 Apr 23.
-
Rodrigues EB, Costa EF, Penha FM, Melo GB, Bottos J, Dib E, et al. The use of vital dyes in ocular surgery. Surv Ophthalmol. 2009 Sep-Oct; 54(5): 576-617.
-
Rodrigues EB, Penha FM, Farah ME, de Paula Fiod Costa E, Maia M, Dib E, et al. Preclinical investigation of the retinal biocompatibility of six novel vital dyes for chromovitrectomy. Retina. 2009 Apr; 29(4): 497-510.
-
Thompson JT, Haritoglu C, Kampik A, Langhals H. Should Indocyanine green should be used to facilitate removal of the internal limiting membrane in macular hole surgery. Surv Ophthalmol. 2009 Jan-Feb; 54(1): 135-8.