REQUESTED COMMUNICATION
Clinical and surgical management of primary congenital glaucoma
MORALES-FERNÁNDEZ L1, MARTÍNEZ-DE-LA-CASA JM2, SAENZ-FRANCES F1, MÉNDEZ C2, GARCÍA-FEIJOO J2
Glaucoma Unit. Hospital Clínico
San Carlos. Madrid
1 Graduate in Medicine.
2 Ph.D. in Medicine.
ABSTRACT
Primary congenital glaucoma (PCG) is an infrequent disease with an estimated incidence of one every 10,000/15,000 live births (world population). However, this incidence seems to be significantly lower in our country (2.85/100,000 live births) (1).
PCG is characterized mainly by iridocorneal angle alterations, specifically trabecular mesh alterations which interfere the normal flow of aqueous humor, leading to increased intraocular pressure (IOP) (2).
Due to the early age at which patients are diagnosed and as occurs in other types of glaucoma, said increased IOP progressively damages the optic nerve. However, due to the high distention capacity of ocular structures in the first years of life the corneal diameters and axial length of the globe increase (3).
The vast majority of said conditions are sporadic although an autosomic recessive inheritance pattern with incomplete penetrance has been described in a high percentage of cases, making it essential to consider the analysis of known genes as well as genetic counseling for affected families (1).
The management (diagnostic and treatment) of primary congenital glaucoma is described below, together with the genetic mutations involved in this pathology.
DIAGNOSTIC AND FOLLOW-UP
The typical triad (tearing, photophobia and blepharospasm) is essential for diagnosing suspected PCG. Said symptoms are not exclusive of this condition and for this reason a differential diagnostic is necessary (4).
The short age of patients and the difficulty to establish the diagnostic and follow-up makes it necessary in the majority of cases to carry out an examination under general anesthesia until the age and cooperation of the patients increase. Said examinations must be carried out in a systematic manner, mainly focusing on the time of the measurements and the type of anesthesia to be applied as this can change the results of some parameters, mainly IOP.
Every examination must include (fig. 1):
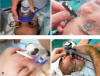
Fig. 1: examination with anesthesia
(a) enables taking biometric measurements and exploring the anterior and
posterior poles (b). the Perkins applanation tonometer is most widely used (c)
although there are useful alternatives such as Icare Pro (d).
-
Corneal diameter measurement, using a strabismus compass. Horizontal and vertical diameters must be measured.
-
Gonioscopy: the angle can be visualized only with adequate corneal transparency (2). At the gonioscopic level the following findings may be made: a) Barkan membrane, a transparent membrane which covers the trabecular mesh; b) Whitish membrane covering the angle, probably an evolution of Barkan’s membrane; c) High iris insertion, close to or virtually at the level of Schwalbe line; and/or d) mesodermic remains.
-
Biometrics. Measuring the axial length is useful for diagnostic as well as for assessing the progression of the disease.
-
Examination of the anterior pole with a surgical microscope to assess possible anterior segment alterations, corneal transparency, Haab striations, appearance of the iris, presence and position of the lens among others (2-4).
-
Ocular fundus examination: enables optic nerve observation to discard any associated retino- choroidal alteration.
-
Tonometry should be carried out at the end of the examination to minimize the effects of anesthetics. It is carried out following the standard procedure with the Perkins-type manual applanation tonometer. In recent years new tonometers have appeared which could be useful in this type of patients, such as the Icare-Pro rebound tonometer or Tonopen (5).
TREATMENT
Treatment of PCG is mainly surgical due to poor response to pharmacogical treatment. In contrast with treatment for adults, medical treatment is reduced to a number of assumptions such as awaiting surgery as a temporary treatment, endeavoring to recover corneal
transparency in the presence of edema which makes goniotomy difficult, or in patients with multiple surgeries in which medical treatment is applied to delay subsequent surgery.
A broad range of surgical techniques are applicable to PCG. However, this paper will outline only the most frequently applied procedures.
Goniotomy
Goniotomy is the most widely applied technique for congenital glaucoma. It was first described in 1938 and has changed very little since then (6). The action mechanism of goniotomy is to open a path for draining the aqueous humor towards Schlemm’s canal in addition to cutting the tissue which sometimes covers the iridocorneal angle. In high iris insertion cases, after goniotomy the iris can descend, broadening the iridocorneal angle.
Adequate visualization of the angle coaxial requires light and a gonioscope. A broad range of gonioscopes is available (Barkan, Koeppe, Worst, Swan, among others). The authors utilized the Worst lens which is held on to the ocular surface by means of 4 anchor points, enabling the surgeon to control ocular globe movements. The Worst lens comprises an inbuilt irrigation system that maintains good anterior chamber visibility throughout the surgery. There is a large variety of goniotomes although the most widely used at present is the Worst hydrostatic goniotome. The goniotome is introduced at the level of the limbus at the opposite end of the area in which the goniotomy will be performed. Approximately 120° are covered and therefore it can be repeated up to 3 times in the same eye (fig. 2).

Fig. 2: Goniotomy: The Worst lens
enables angle visualization (a) and anterior chamber access (b). After a section
of up to 120 degrees using the goniotome (c) the section is closed with simple
stitch (d).
The success of conventional goniotomy varies according to each studied series although the control rate varies between 33% and 100%. The largest published series were those of Bietti (7) and Shaffer (8) which reported a control percentage between 68% and 77%. Shaffer studied 205 eyes intervened with goniotomy and obtained a control percentage of 77%. For Shaffer the results are much better (control of 90%) when diagnosed between birth and age 2 but much worse when diagnosed beyond this period (30% control).
Without a doubt, the most frequent complication of goniotomy is angle bleeding during the intervention and the ensuing post-surgery hyphema which in most cases is small and resolves in a few days. Other less frequent but more severe complications are lens anterior capsule rupture or accidental incision of Descemet’s membrane with the goniotome.
Trabeculectomy
In CG trabeculectomy is not very different than the same procedure in adults. The surgical technique is as follows (fig. 3): a conjunctival flap is made based on the fornix (although some authors continue to prefer the classic opening based on the limbus) followed by 2 vertical sclerotomies that are joined through a horizontal approximately 1 mm in front of the insertion of the superior rectus. The superior mat is dissected at a depth of 2/3 overall thickness of the sclera and is continued until 2 mm of the cornea have been penetrated. The deep mat is carved with vannas or small scalpel and subsequently a base and broad iridectomy is performed. The sclera is closed by means of Nylon 10/0 stitching and the conjunctiva with Vicryl 8/0 (9).
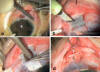
Fig. 3: Trabeculectomy: After
dissection of the conjunctiva (a) the surface mat is carved (b). After
completing the deep mat and the iridectomy (c), the surgery is completed closing
the sclera and conjunctiva (d).
In what concerns adult glaucoma, the main difference the authors found was the extreme thinness of the sclera which makes surgery considerably more difficult. Likewise, the distention of the sclerocorneal limbus in these patients requires deeper penetration in the cornea to ensure that the trabeculum is included. It is important to consider the high probability of re-interventions in these cases and for this reason it is convenient to displace nasally the trabeculectomy to leave sufficient space for a second trabeculectomy if necessary.
Several authors such as Dureau (9) reported trabeculectomy success rates in this group of patients of 69% after a single surgery and a follow-up of 57 months. In many cases the authors found it necessary to perform a second trabeculectomy or even 3 or 4, which were carried out in the inferior quadrant even though it is an unusual position (10,11) (a controversial position according to some authors). It is important to emphasize that age is a crucial prognostic factor: the younger the age of the first surgery the lower will be the success rate and the less encouraging will be the prognosis (9).
Trabeculectomy complications are more frequent and more severe than those associated to the technique described above (goniotomy). The appearance of hypotony, choroidal hemorrhage, vitreous hemorrhage due to posterior lens rupture and even endophthalmitis is more frequent than in goniotomy. For these reasons, most authors do not propose trabeculectomy as the first choice.
Valve implants
Valve implants are reserved for patients who could not be controlled despite several surgeries. Molteno was the first to use these implants in PCG. At present there are several valve types (Ahmed, Baerveldt and others). However, the model of choice is Ahmed’s valve, comprising a silastic tube connected to silicone membranes which act as a valve and are joined to a 185mm2 polypropylene platform. The valve is designed to allow the exit of aqueous humor when IOP exceeds 8 mmHg.
The surgical technique is similar to that applied in adult surgery with a few exceptions. Similarly, the upper temporal quadrant is preferred. After dissecting the conjunctiva and tenon, purging is performed and the adequate operation of the valve is verified. The body of the valve is placed between the lateral and upper rectus, and fixed at 8-9 mm of the limbus with nylon 9/0. After the length of the tube has been determined it is cut with a beveled section and introduced in the anterior or posterior chamber through the sclera at 1 mm of the limbus. After covering the path of the exterior tube with scleral patch, the conjunctiva is closed with continuous stitching.
Published studies report variable success rates, ranging from 90% to 70% of cases after 12 months follow-up (12,13), with significant reductions at 24 months, while the results range between 58% and 64% (12,13). However, if the results of these implants are evaluated throughout a five-year term, the success rates diminish to about 33%, although they increase to 69% if a second implant is considered (14). The use of mitomycin C should be considered in valve surgery in the same way it is used as in adult surgery (15). Even so, the results seem to be contradictory because the success rates are lower when this substance is applied. This could be due to increased fibrosis around the device in this group of patients.
Among the complications in valvular implants, those secondary to the tube are particularly relevant, i.e., poor position close to the lens of the endothelium, anomalous path direction or length or obstructions thereof. Further complications include perivalvular fibrosis, implant extrusion or diplopia.
GENETIC STUDY OF PRIMARY CONGENITAL GLAUCOMA
It was considered that most PCG cases were sporadic. However, recent studies report a recessive autosomic inheritance pattern with incomplete penetrance and variable expressiveness, with gene CYP1B1 mutations being most frequently associated (16).
There are 3 genetic loci that seem to be perfectly related to PCG: GLC3A (2p21) and GLC3B (1p36) (through mapping, this region was located in 1p36.2-1p36.1 adjacent to the loci for neuroblastoma and Charcot-Mare-Tooth type 2A) and GLC3C (14q24.3) (17). Even though these 3 loci are broadly referenced in the literature, to date only CYP1B1 has been related to locus GLC3A. Accordingly, the second and third loci correspond to unidentified genes.
Gene CYP450 is considered to be the main genetic factor involved in PCG. The mutation frequency accounts for up to 85% of cases. CYP1B1 is part of the superfamily of cytochrome P450 which synthesizes a protein with catalytic activity expressed in the eye of the fetus and the adult, particularly in the iris, the trabecular mesh and the ciliary body, which accounts for its involvement in PCG (18,1).
Even though the CYP1B1 mutations have also been described in other forms of early onset glaucoma associated to anterior segment congenital anomalies, these are not considered to be PCG, as well as the Rieger or Peters anomalies. The possible relationship of gene MYOC (myocilin) with gene FOXC1 (forkhead box C1) with PCG is worthy of special mention. These have been mapped in chromosomes 1q21-q31 and 6p25 respectively.
To conclude, considering the high association of gene CYP1B1 with PCG and the potential severity of the condition, at present the authors consider it advisable to carry out a detailed genetic study of the family and to prepare adequate genetic counseling.
REFERENCES
-
CYP 1B1 mutations in Spanish patients with primary congenital glaucoma: phenotypic and functional variability. Campos- Mollo E, López-Garrido MP, Blanco-Marchite C, Garcia-Feijoó J, Peralta J, Belmonte-Martinez J, Ayuso C, Escribano J. Mol Vis. 2009; 15: 417-31.
-
Allen L, Burion HM, Braley AE. A new concept of development of anterior chamber angle: Its relationship to developmental glaucoma an other structural anomalies. Arch Ophthalmol 1955; 53, 783-98.
-
Anderson DR. The development of the trabecular meshwork and its abnormality in primary infantile glaucoma. Am Ophthamol 1981; 79, 458-485.
-
Kupfer C, Kaiser-Kupfer M. Observations on the development of the anterior chamber angle with reference to the patogenesis of congenital glaucomas. Am J Ophthalmol 1979; 88, 424-26.
-
Martinez-de-la-Casa JM, Garcia-Feijoo J, Saenz-Frances F, Vizzeri G, Fernandez-Vidal A, Mendez-Hernandez C, Garcia-Sanchez J. Comparison of rebound tonometer and Goldmann handheld applanation tonometer in congenital glaucoma. J Glaucoma. 2009 Jan; 18(1): 49-52.
-
Shaffer RN. Goniotomy technique in congenital glaucoma. Am J Ophthalmol. 1959 May; 47(5, Part 2): 90-7.
-
Bietti GB, Quaranta CA. Indications for, and results of the operation of incision of the iridocorneal angle (goniotomy, goniotrabeculotomy or trabeculotomy). Vestn Oftalmol. 1968 May-Jun; 81(3): 9-21.
-
Shaffer RN. Prognosis of goniotomy in primary infantile glaucoma (trabeculodysgenesis). Trans Am Ophthalmol Soc. 1982; 80: 321-5.
-
Dureau P, Dollfus H, Cassegrain C, Dufier JL. Long-term results of trabeculectomy for congenital glaucoma. J Pediatr Ophthalmol Strabismus. 1998 Jul-Aug; 35(4): 198-202.
-
Mandal AK, Gothwal VK, Bagga H, Nutheti R, Mansoori T. Outcome of surgery on infants younger than 1 month with congenital glaucoma. Ophthalmology. 2003 Oct; 110(10): 1909-15.
-
Kothari M, Balankhe S. Inferior trabeculotomy and trabeculectomyfor refractory pediatric glaucomas. Indian J Ophthalmol. 2011 Jul-Aug; 59(4): 323.
-
Djodeyre MR, Peralta Calvo J, Abelairas Gomez J. Clinical evaluation and risk factors of time to failure of Ahmed Glaucoma Valve implant in pediatric patients. Ophthalmology. 2001 Mar; 108(3): 614-20.
-
Englert JA, Freedman SF, Cox TA. The Ahmed valve in refractory pediatric glaucoma. Am J Ophthalmol. 1999 Jan; 127(1): 34-42.
-
Ou Y, Yu F, Law SK, Coleman AL, Caprioli J.Outcomes of Ahmed glaucoma valve implantation in children with primary congenital glaucoma. Arch Ophthalmol. 2009 Nov; 127(11): 1436-41.
-
Al-Mobarak F, Khan AO.Two-year survival of Ahmed valve implantation in the first 2 years of life with and without intraoperative mitomycin-C. Ophthalmology. 2009 Oct; 116(10): 1862-5.
-
Vincent AL, Billingsley G, Buys Y, Levin AV, Priston M, Trope G, et al. Digenic inheritance of early-onset glaucoma: CYP1B1, a potential modifier gene. Am J Hum Genet 2002; 70: 448-60.
-
Batiyeva S, Marfany G, Gonzalez-Angulo O, Gonzalez-Duarte R. Mutational screening of CYP1B1 in Turkish PCG families and functional analyses of newly detected mutations. Mol Vis 2007; 13: 1458-68.
-
Stoilov IR, Costa VP, Vasconcellos JP, Melo MB, Betinjane AJ, Carani JC, Oltrongge EV, Sarfarazi M. Molecular genetics of primary congenital glaucoma in Brazil. Invest Ophthalmol Vis Sci 2002; 43: 1820-7.