Cruces Hospital. Vizcaya, Spain.
Experimental Ophthalmology Group (GOBE), Basque Country University (UPV/EHU).
1 Graduate in Medicine. Ophthalmology Service.
2 Graduate in Medicine. Pediatric Service.
3 PhD in Medicine. Ophthalmology Service.
INTRODUCTION
In addition to housing the ocular globe, the orbit comprises some soft parts which are vulnerable to inflammation or infection. Preseptal cellulitis (PC) affects the tissues in the anterior locations from the orbitary septum, while orbital cellulitis (OC) involves intraorbitary fat and muscles. The most frequent cause is extension due to continuity of the adjacent paranasal sinus infections. This is a frequent pathology in the pediatric age which must not be underestimated due to its life-threatening potential. This paper is a review of the main demographic, microbiologic, diagnostic and therapeutic aspects of this pathological entity, comprising the main articles published since 1985, when the vaccine against Haemophilus influenzae type b (Hib) became universal in the Western world and contributed to the modification of the microbiologic spectrum involved in the etiology of PC and OC.
ANATOMY
The orbit constitutes a non-expandable bone cavity which houses the ocular globe, the most anterior portion of the optic nerve and the extraocular muscles as well as orbitary fat. It is closed anteriorly by the eyelids and their connective tissue.
The differentiation between OC and PC is based on the location of the inflammatory-infectious process according to an anatomical structure that is the orbitary septum, made up by a fibrous, thin and mobile film which joins the orbitary edge with the peripheral edge of the tharsus, forming the arcus marginalis (1,2). It functions as a barrier against inflammatory processes involving the eyelids and surrounding areas that prevents their penetration into the orbit (3,4).
The orbit can be divided in 3 «surgical spaces»: the intracone, extracone and subperiostium spaces. One or more of these spaces can lodge a pathological process in an OC. The intra-and extraconal spaces are defined by the muscular fascial septum that constitutes the orbitary cone. In the posterior area, close to Zinn’s ring, this septum is incomplete, thus facilitating the extension of an inflammation from one space to the other. However, the sub-periostium space is virtual, although it frequently becomes real in OC. The periostium is firmly inserted in the skeleton along the bone sutures and the orbitary edge, although there are some lax areas between the insertions where abscesses can be formed.
Three of the four orbit walls form part of the paranasal sinuses. The medial wall forms part of the ethmoidal sinus, the inferior wall is part of the maxillary sinus and the superior wall is part of the frontal sinus. Both the maxillary and the ethmoidal sinus are completely formed at the time of birth and the pneumatization of the frontal sinus is completed only in the fifth or sixth year of life. The first two sinuses have the peculiarity of being separated from the obit by a thin layer of bone which, in the case of the ethmoidal sinus, presents perforations for vessels and nerves which could facilitate the intra-orbitary extension of a sinus infection.
The characteristic avalvular vascularization of the orbit that passes through the cavernous sinus could involve this space with the ensuing risk of intracranial extension through the adjacent meninges.
![]()
Fig. 1: images of a 2-year-old child
affected by orbital cellulitis. A: medial subperiostium abscess (arrow) in the
right orbits accompanied by ethmoidal sinusitis (asterisk). B: image evidencing
the resolution of the condition after IV treatment with amoxicillin-clavulanic
and transnasal sinus drainage.
EPIDEMIOLOGY
PC and OC are more frequent in children than in adults. In turn, PC expresses in smaller children and is up to 3 times more frequent than OC (3).
Articles published to date agree on the direct relationship between the patient age and the severity of the process, with PC being more frequent in children under 5 and OC in children over that age (5).
In a review of 315 pediatric patients with both types of infections, 94% were PC. The mean age of this Boston group was of 3 for PC and 5 for OC (6).
In a study published by La Orden Izquierdo et al which included 66 children, the mean age for PC was 38 months (7). Another group, led by Rodríguez Ferrán et al that reviewed 107 cases found a mean of 52 months (8).
On the other hand, in a review of 38 children diagnosed with OC carried out by McKinley et al the mean age was of 6.8 years (9). In turn, the group of Seltz identified a mean of 6 years in a review of 94 children who were also diagnosed with OC (10).
This type of orbitary infection is more frequent in males (10-13). It usually expresses in winter and unilaterally (14,15).
ETIOPATHOGENY
The most frequent cause of cellulitis is a bacterian infection, which may originate by direct extension from an adjacent sinusitis site, direct innoculation after traumatism or skin infection, or bacteriemic infection from a distant site (16,17).
In the absence of periorbital traumatism or local infection, at least 75% of OC are due to sinusitis, with the ethmoidal sinus being the most frequent source of infection (16,18-20). In a series of 315 children collected by Ambati et al in 2000. 44/297 cases of PC and 18/18 of OC were identified as originating in a sinus infection (6).
The existence of common avalvular venous drainage between the paranasal sinuses and the orbit, together with neurovascular orifices in the wall of the ethmoidal sinus (that are compromised when the sinus mucosa is edematized) facilitate the entry of the microorganisms that cause sinusitis into the orbit (16,18).
MICROBIOLOGY
A turning point can be identified in the microbiology of OC, i.e., the standardization of the vaccine against Hib in 1985. Until that year, this highly active germ that causes meningitis and bacteremias was the first cause of OC (21). Ambati et al demonstrated not only a reduction in the prevalence of cellulitis due to Hib but also suggested diminished overall prevalence of cellulitis. This is explained by recognizing this germ as a facilitator for the access of other organisms in the orbit as joint pathogens (6). At present this must still be considered in children when adequate immunization is doubtful.
In Spain the vaccine against Hib was made compulsory in 1998 and since then it is included in the vaccination calendar. Children at the age of 2, 4, 6 and 18 months of life are administered immunization in all the autonomous communities of the country.
There is no consensus concerning the most frequently responsible microorganism although all authors seem to agree that the absence of traumatism is treated in 100% of gram-positive cocci cases.
Several authors have published that Staphylococcus aureus is the most predominant microorganism (9,13,22,23). Others refer to Streptococcus pneumoniae (24-26). The team of Brook as well as that of Rudloe have identified Streptococcus viridans as the most frequent pathogen (11,18). At present there is an increasing number of authors who emphasize the leading role being played by the group of Streptococcus anginosus (10.23).
Obviously, there is a debate regarding the identification of the most frequent microorganism. Even so, many authors point at Streptococcus pyogenes as one of the most frequent ones (10,11,18,23).
In areas where methicillin-resistant Staphylococcus aureus (MRSA) is prevalent, isolated cases of cellulitis caused by this pathogen have been identified (9,11,27).
In a patient with suspected PC or OC, blood samples should be taken either from abscess or sinus material whenever possible. Before establishing immunization against Hib in vaccination calendars for children in the Western world, hemocultures were usually positive while at present they are negative, above all in older children (3,9,11,22). The poor performance of hemocultures was evidenced by Rudloe et al with 3/407 positive hemocultures and Seltz et al with only 3% positive (10.11). However, in children undergoing surgical approaches in which direct samples are obtained, a pathogen is identified in up to 81% of cases (10).
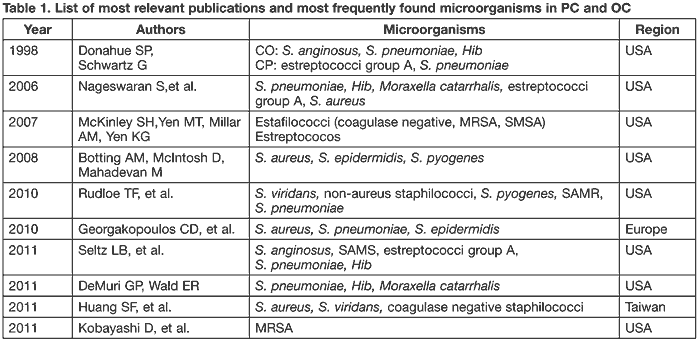
Table 1 summarizes the most frequent microorganisms according to the most relevant authors.
CLINIC
Due to the therapeutic and prognostic implications, it is extremely important to differentiate PC from OC in our patients.
PC expresses with palpebral edema which can achieve a degree of intensity that prevents the spontaneous opening of the eye, and eyelid erythema. In general, the ocular globe is not involved and neither is pupil reactivity, visual acuity or motility. Ocular movements do not cause pain and there is no chemosis.
OC is associated to fever, proptosis, chemosis, ophthalmoplegy associated or not to diplopia and pain with ocular globe movements. Diminished visual acuity and pupil alterations indicate involvement of the orbital vertex.
According to Sobol et al, ophthalmoplegy and proptosis are a positive predictive value of 90% for orbital infection. Their absence implies a negative predictive value of 93% (28).
Retrospective studies carried out to date endeavor to provide clinical guidelines for the differential diagnostic between both entities.
Botting et al include a list of 262 cases of children in which proptosis was observed in 94% of patients with OC, against 0.9% who did not exhibit proptosis. Diplopia (54% against 0.9%) and ophthalmoplegy (11% against 0.4%) also seem to be linked to the involvement of intra-orbitary structures (23).
Fever (a temperature > 38ºC) and leukociytosis (total > 15,000 cells/mm3) are less common in limited cases but can appear in children with both processes.
Recurrence is infrequent and usually due to underlying causes such as sinus anomalies (29).
COMPLICATIONS
Complications are more frequent in OC cases although can also be observed in extended PC cases. According to retrospective series, the most frequent abscess is the subperiostium abscess (59%) followed by the orbitary abscess (24%) (10,23,26,28,30).
In a series of 298 patients, an orbital abscess was determined only in 2% of the cases in which imaging tests were carried out (11).
Other observed complications include diminished visual acuity, cavernous sinus thrombosis, brain abscesses, central retinal artery obstruction and even death. Osguthorpe and Hochman described up to 11% of cases of diminished visual acuity and up to 2% of deaths due to dissemination of the infectious process in North American children with OC (31).
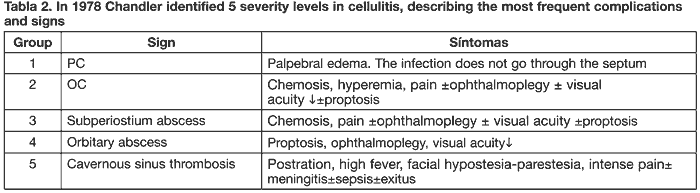
In 1978, Chandler classified potential pediatric age cellulitis complications in 5 severity levels, which are shown in Table 2 (16).
Subperiostium non-medial abscesses tend to appear in older children and this explains why children under 9 respond better to isolated antibiotic therapy whereas surgery is frequently required in older children (30).
Orbitary abscess compromises the optic nerve and vessels, and this translates into diminished visual acuity which could be irreversible.
Cavernous sinus thrombosis, caused by the dissemination of the infection through the avalvular veins, requires urgent hospital management due to its life threatening potential.
DIAGNOSTIC
The diagnostic of this pathology is mainly clinic, with anamnesis and physical assessment being its main pillars. Image tests may be necessary although their usefulness is controversial because the pediatric age is the main age affected by this pathology and in this period of life radiation should be avoided according to the ALARA criteria, based on applying minimum radiation as reasonably necessary for carrying out tests.
In what concerns image tests, nuclear magnetic resonance (NRM) produces better resolution for diagnosing soft tissue involvement, although it is not always available. For this reason, computerized tomography (CT) is more widely used.
Criteria for indicating imaging tests have been defined. These are proptosis, ophthalmoplegy, pain with movement, diplopia, loss of vision, edema, neutrophilia exceeding 10,000 cells/mm3, central nervous system involvement, infants under one year old or difficult to explore, absence of improvement after 24-48 hours of antibiotic treatment or worsening (11,28,32). The existence of at least one of these conditions would justify image tests for a child.
In 2010, Rudloe et al carried out a study in which 12% of patients submitted to CT exhibited significant findings. The involvement of the maxillary and ethmoidal sinuses was the most frequent finding, appearing respectively in 86% and 84% of cases. Multiple sinus involvement was identified in 80% of cases. In addition, it was evident that 50.5% of orbital abscess cases identified by CT did not refer clinic related to ophthalmoplegy, proptosis or pain with movement. Accordingly it can be inferred that vigilance must always be maintained despite low-profile clinics (11).
The early diagnostic of OC complications is very important for the visual prognosis of our patients. As pointed out by Uzcátegui et al up to 10% of patients may lose their eyesight. In a series of 111 children where the findings were identified in the imaging tests, 2 lost their eyesight (34).
The group of Vu et al recommended the use of NMR when intracranial complications are suspected, such as empyema or cavernous sinus thrombosis, due to its higher resolution (33).
Being aware of the existence of post-gadolinium kidney fibrosis, the American group of Sepahdari et al researched the role of DWI (Diffusion-Weighted Imaging), a technique that was previously utilized in orbitary pathologies, against the conventional use of gadolinium in the differential diagnostic between orbital cellulitis and orbital abscess. In a series of 9 cases, they verified the presence of supuration complications in 6 cases, 2 of which were pediatric patients. They concluded that the use of DWI increased confidence in the orbital abscess diagnostic and in addition it was a fast technique and therefore propose its inclusion in all the routine intracranial NMR studies. However, DWI was not able to displace the use of gadolinium although its application should be considered in patients with diagnosed kidney insufficiency to avoid aggravating said pathology (35-39).
MEDICAL TREATMENT
Medical therapy is based on broad spectrum antibiotics focused on treating not only cellulitis but also predisposing factors such as sinusitis when present. To date, antibiotic therapy developments have reduced the morbidity and mortality typically associated to this pathology (40). It is important to know the local flora that is involved because pathogens and their resistance to antibiotics vary depending on the geographic area (3).
Both for PC and OC, several authors propose associating treatment focused on sinusitis if present. The indication is for intranasal corticoids to diminish the mucosa edema and facilitate drainage, together with nasal irrigation (41).
The use of intravenous corticoids is controversial because, even though it keeps the inflammatory reaction in check, it could be dangerous because it generates secondary immunosuppression (3,7,42,43).
Preseptal celullitis
In what concerns selected antibiotics and due to the poor performance of hemocultures, empirical therapies are prescribed against the above-mentioned most frequently found microorganisms (3). In addition, to date there are no randomized studies.
In children over one year of age without associated systemic problems, broad range oral antibiotic treatment is established at an early stage with close patient follow-up. If there are no improvement signs within 24 hours or if the condition of the patient worsens the oral pathway is substituted by IV. The possibility of resistant microorganisms must always be taken into account in these cases (3).
In contrast with the above, the group of La Orden Izquierdo et al proposed initial IV amoxicillin-clavulanic acid in PC associated or not to IV corticoids in any child. In addition said group proposes topical antibiotic treatment. This appears to be a controversial issue (7).
The most widely used antibiotics are vancomycin, amoxicillin-clavulanic acid, cefotaxime and ceftriaxone. Treatment is maintained between 7 and 10 days up to the complete disappearance of signs and symptoms (3).
Initial parenteral therapy associated to hospital admittance is indicated in patients under one year of age, children who are not vaccinated against Hib and S. pneumoniae, who are immunosuppressed and who exhibit evidence of greater severity or systemic toxicity.
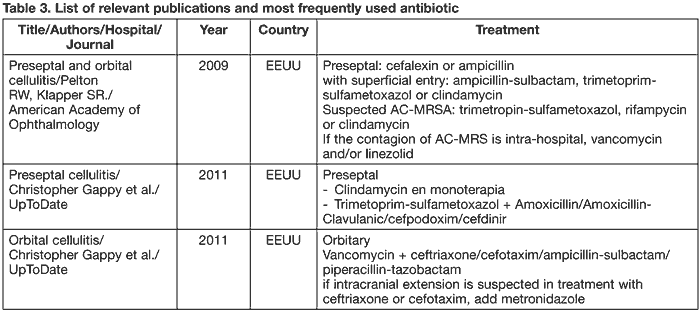
Table 3 illustrates different therapeutic guidelines.
Orbital cellulitis
Due to the significant number of possible complications including death, intravenous treatment should be initiated at an early stage (20).
The majority of patients exhibit good response to antibiotic treatment and do not require surgical approach.
Generally, in order to achieve broad coverage of gram-positive, gram-negative and anaerobic cocci cefotaxime and metronidazole or clindamycin are used simultaneously. Other options are piperacillin-tazobactam, tiarcillin-clavulanic and ceftriaxone. In penicillin-allergic patients a combination of vancomycin and fluoroquinolone can be used (3).
Cannon et al propose treating OC with oral antibiotics on the grounds that ciprofloxacine and clindamycin in association have a similar bioequivalence in the oral and parenteral pathway and oral treatment would avoid the side effects of IV access. Although the number of cases in their study is small (only 36 patients), the results are significant and it would be interesting to carry out further research in this area (19).
There is no consensus on the duration of the IV treatment before switching to the oral pathway, but it seems reasonable to wait until the patient is free of fever and exhibits a substantial improvement of the orbitary signs (39).
THE CRUCES HOSPITAL PROTOCOL
Table 4 illustrates the action algorithm of the Cruces University Hospital.
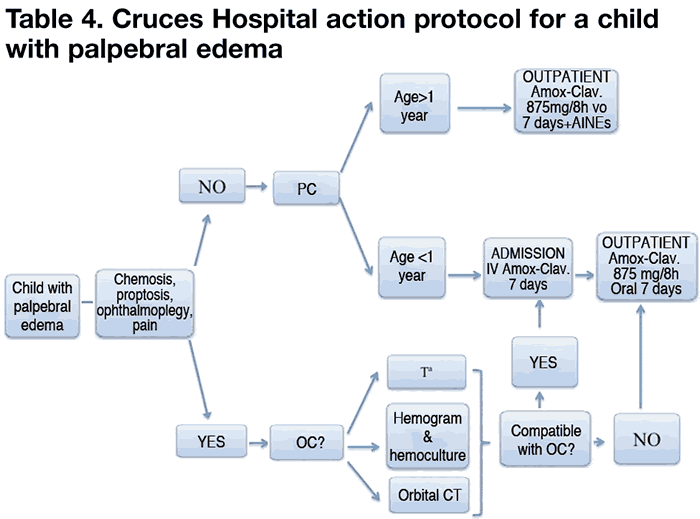
In contrast with the majority of publications referenced above, in our hospital the antibiotic of choice is amoxicillin-clavulanic acid, with ceftriaxone being the second choice.
Children suspected with PC or OC are received at the Pediatric Emergency Service of the Cruces University Hospital. After a general exploration, the pediatricians refer the patient to the Ophthalmology Emergency Service of the same hospital for a specific assessment of the ophthalmological signs and symptoms. The children who must be admitted for treatment and control of the evolution of their condition are under the Pediatrics Service. The Ophthalmology Service performs daily assessments of visual acuity, ocular movements, pupil reaction and anterior pole and ocular fundus exploration while the patient remains in the hospital. The patient release and subsequent outpatient control is decided jointly after the fever and ophthalmological signs have resolved.
If an orbitary and/or subperiostium abscess needs to be drained, this intervention is carried out by an orbit and oculoplasty specialist. Sinus drainage is performed by the ENT Service.
SURGICAL TREATMENT
Surgical interventions are indicated in OC cases which associate foreign bodies and others which are associated to orbitary and/or subperiostium abscesses, but the fact remains that there are no universally accepted protocols (20,32).
Some surgeons have proposed an immediate drainage of abscesses while others prefer observation (20,32,44).
Medial subperiostium abscesses in children usually respond positively to antibiotics and therefore surgical drainage is an infrequent option (45,46). In a series published by Ryan et al comprising 68 children with subperiostium access, only 21 required surgery (30).
According to Harris, subperiostium abscesses can be observed unless one of the following criteria is present: age 9 or more, frontal sinusitis, non-medial subperiostium abscess, suspected anaerobic infection, recurrence after surgical drainage, chronic sinusitis or optic nerve or retina compromise (45).
Todman’s group established abscess size as a fundamental criteria for indicating surgery, so that patients with abscesses exceeding 10 mm diameter or having the volume over 1,250 mm3 are drained (47).
As regards surgical techniques, the approach can be external or transcaruncular endoscopic (48). The choice of approach would be mainly determined by the skill of the surgeon and/or the location of the subperiostium abscess.
In the case of orbitary abscesses, which appear in up to 24% of OC cases according to Rudloe et al, surgical approach is generally required because these abscesses frequently associate optic nerve compromise and risk of intracranial complications (11,20.32).
In some cases it is necessary to drain the affected sinuses in order to control the infection. Debridling is only required in patients exhibiting destructive sinusitis. Therefore, in some occasions, multidisciplinary cooperation between ophthalmologists and ENT specialists is essential. The material drained from an abscess as well as a sinus must be sent for culture due to its high reproducibility in order to choose the most effective antibiotic therapy.
The management of intracranial complications requires admission into the intensive care unit, strong intravenous antibiotic treatment and control by a neurosurgeon. However, intensive therapy does not insure patient survival due to the high mortality rate of this condition.
CONCLUSIONS
Cellulitis are classified as preseptal or orbital depending on the location of the infectious-inflammatory site, taking the orbitary septum as reference. Cellulitis is more frequent in the pediatric age and the most frequent etiology is extension due to continuity of paranasal sinus infection. Even though there is no consensus about the most frequent microorganism, gram-positive cocci are the most frequently found cause. Clinic with palpebral edema is similar in both, although typically OC is characterized by ophthalmoplegy, proptosis and pain upon ocular movements. In addition this is associated to a higher risk of complications and therefore a precise diagnostic supported by image tests must be made as soon as possible to initiate oral or IV antibiotic treatment to minimize vital risks.